Energy in Biology
Have you ever felt a “sugar rush” that made you jump off the walls like crazy, only to “crash” later? Or felt really grumpy and tired, only to immediately feel like your normal self again after eating a snack? That’s biology at work! As we’ve mentioned a few times already in this class, and as you probably already know from your own intuition, our bodies need energy to survive. We’ve seen some ways that our bodies use energy, too: turning on certain enzymes, for example, or powering active transport of important nutrients, or making new proteins. But what is energy from a biological perspective, anyways? Where does it come from? And what do we really mean when we say that energy “powers” a cell?
What is Energy?
Energy is defined as the power to do work. It’s not matter, or something we can physically touch, so it can be a little weird to think about sometimes. Pushing a block across the floor—doing work—requires energy. Doing homework requires energy. All the work that a cell does comes down to a chemical reaction, so, in biology, we use energy to “power” chemical reactions. By that, we mean we’re giving them an incentive to happen: All chemical reactions need a little bit of energy to happen, and a lot of the complex chemical reactions that happen in our bodies—like anytime we’re building something—need a lot of energy to happen. So, energy has to come from somewhere.
Where Does Earth's Energy Come From?
|
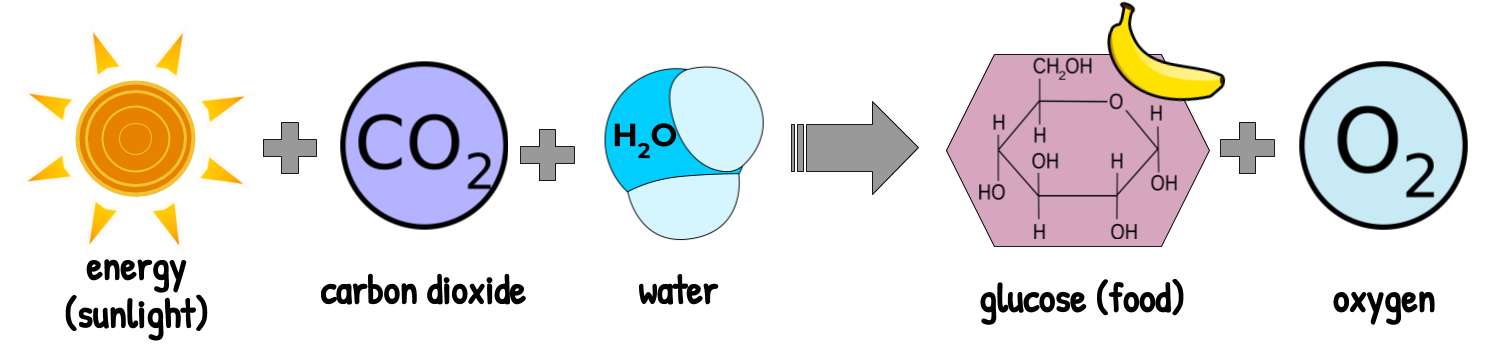
On earth, all of our energy ultimately comes from the sun, which is basically just a really massive nuclear reactor. But, obviously we can’t just power your computer or your body by setting it out in the sunshine all day long. We need some way to convert that energy into something we can actually use. Solar panels take that energy and convert it into a form that can be used to power electronics. Photosynthesis, mostly in plants, takes that energy and converts it into a form that can be used in biology. Photosynthesis is actually an extremely efficient way to harvest the sun’s energy, and it’s a lot better than our best solar panels.
|
If you’d like to learn more about how Earth gets and uses energy, this video gives a great overview:
How Does Photosynthesis Store Energy?
Since biology is just molecules, all biological energy is stored in molecules. When we say we’re storing energy, we mean that, right now, we’re not doing anything with that energy, but we could do something with that energy. We call this potential energy. It’s like when someone tells you that you have the “potential” to be an amazing scientist, or engineer, or musician, or whatever it is that you’re good at. You’re not currently doing that work, but you could do that work, one day. You already know that you get energy out of breaking chemical bonds in a chemical reaction. This is what makes that reaction happen. So, then, that bond must have been storing energy. Ergo, molecules store energy. (Some more than others).
Photosynthesis uses the sun’s energy to turn little molecules—carbon dioxide—into bigger molecules—glucose. Glucose has a lot of bonds, so it has a lot of potential energy. It’s kinda like a battery we can eat.
How Do We Use That Energy?
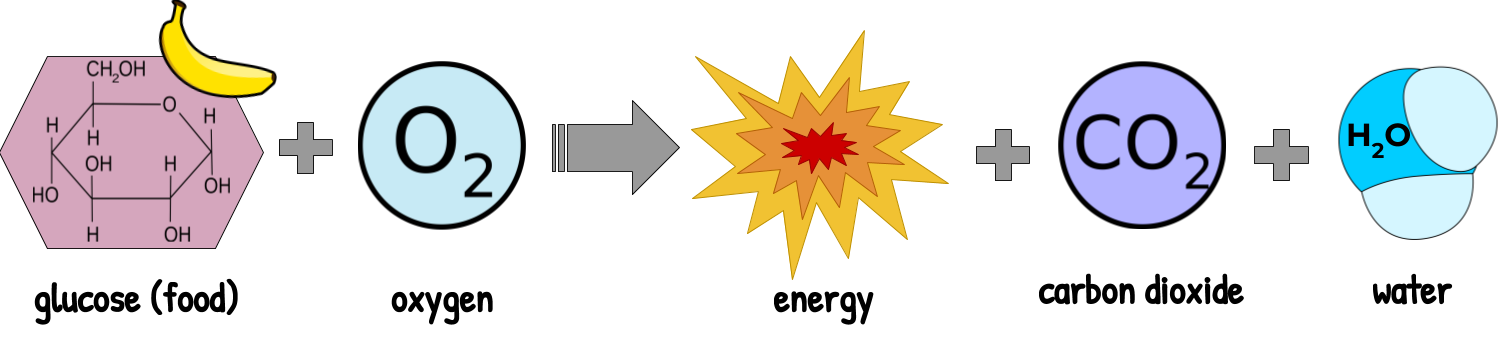
Our bodies break down the bonds in glucose to make new energy. This is called cell respiration. The more useful form of energy that we make from cell respiration is used to power other reactions in the cell, like making new proteins or telling those proteins how to behave.
You already know that glucose stores a lot of energy. If you need a reminder, check out this video:
In this video, the chemical potential energy stored in the bonds of glucose is turned into heat and light energy in the form of an explosion.
Heat and light energy is, however, not that useful for our bodies. If it were, we’d just get what we need from the sun. So, we need to be able to turn the sugar energy into a form that is immediately useful for our bodies. But what form would biologically useful energy take? Batteries? Plugging you into an outlet? Setting you on fire? Nope, it’s another molecule.
Heat and light energy is, however, not that useful for our bodies. If it were, we’d just get what we need from the sun. So, we need to be able to turn the sugar energy into a form that is immediately useful for our bodies. But what form would biologically useful energy take? Batteries? Plugging you into an outlet? Setting you on fire? Nope, it’s another molecule.
Adenosine Triphosphate: The Source of All Biochemical Power
All biology is molecules, and all the work that we want to do is some type of chemical reaction. Chemical reactions get “powered” when one high-energy bond is broken and, with that energy, new, lower-energy bonds are formed. So, really, biological energy is not so much “power” and “energy” like “boom, boom, flash of light and some magic” as it is “power” like “I have given you the capacity to achieve what you already wanted—stability.” Sooo… it’s just chemistry.
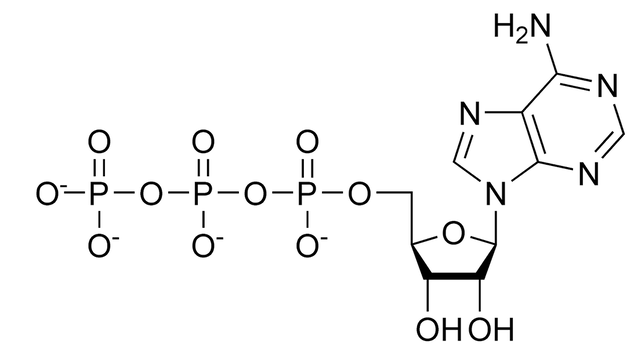
If chemical reactions are powered by breaking high-energy bonds so that more stable ones can be made, the most useful form of “power” or “energy” in a cell is going to be a ridiculously high-energy, unstable molecule. Such a ridiculously unstable molecule would be able to react with just about anything and make all kinds of chemical reactions happen because, even if we’re not making something particularly stable, at least we’re getting rid of this ridiculously unstable bond. Enter our friend Adenosine Triphosphate (ATP):
If chemical reactions are powered by breaking high-energy bonds so that more stable ones can be made, the most useful form of “power” or “energy” in a cell is going to be a ridiculously high-energy, unstable molecule. Such a ridiculously unstable molecule would be able to react with just about anything and make all kinds of chemical reactions happen because, even if we’re not making something particularly stable, at least we’re getting rid of this ridiculously unstable bond. Enter our friend Adenosine Triphosphate (ATP):
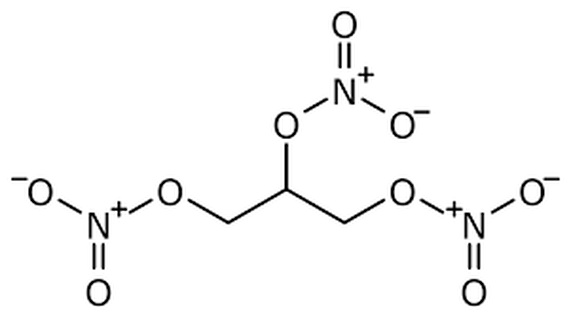
I mean, if a molecule get’s its stability from evenly spreading out charge, making sure everyone has their fair share, and never creating any conflict, like, for example, putting a bunch of negative charges right next to each other even though like charges hate being near each other (opposites attract, and likes repel), then this molecule just seems like a bad idea. I mean, we have three! negatively charged phosphate groups all right next to each other! Held together by O—P bonds, which (you’ll at some point learn) are not particularly stable bonds! I mean, even actual dynamite has the decency to separate its three negative charges with some carbons:
So, basically ATP is just dying to get rid of its third phosphate group to just get rid of some of that extra energy. It wants to be lower energy and stable, just like anything else. So, it will react with just about anything in order to get rid of that phosphate. And, once we’ve put a phosphate on something else, now that molecule is more likely to react, and the next molecule is more likely to react, and so on and so on until what may have been at first a very unlikely reaction—such as taking some relatively stable amino acids and forcing them into a giant protein—is suddenly really eager to get a move on.
This video gives an awesome summary of ATP, our biological energy molecule:
This video gives an awesome summary of ATP, our biological energy molecule:
Summary
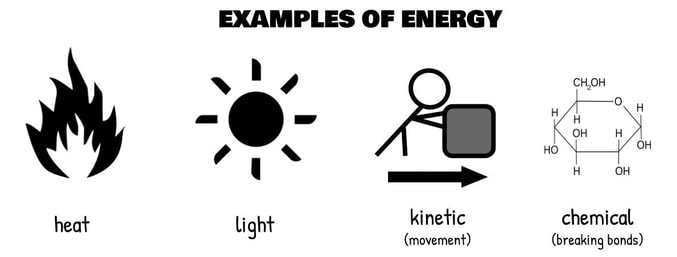
Energy can come in many forms, including heat, light, kinetic energy (movement), sound, and chemical bonds. Energy is never created or destroyed in the universe—this is the first law of thermodynamics. So, energy is constantly just changing forms. New atoms are formed from other atoms at the center of the sun, releasing energy. Heat and light radiation from the sun reaches plants, which turn that energy into chemical energy stored in the bonds of glucose. That glucose is turned into an even more immediately useful form of chemical energy, stored in the bonds of ATP. ATP is so high energy that it will react with almost anything, transferring that energy in the form of a phosphate bond through a reaction until that reaction doesn’t need phosphate to power it anymore. It’s just one giant flow of energy, and the biologically useful forms come packaged as molecules.
You should understand:
You should understand:
- That the Earth’s energy ultimately comes from the sun.
- That plants convert solar energy into chemical potential energy in the form of glucose.
- That all living things convert chemical potential energy in the form of glucose into chemical potential energy in the form of ATP.
- That ATP is an extremely high-energy molecule that will react with almost anything, making all kinds of unlikely chemical reactions in the cell possible. It is the most immediately useful form of stored energy in the cell.
Learning Activity
Content contributors: Emma Moulton