The Structure of Atoms
How small is an atom?
You learned in the last part of this lesson that atoms are really, really small. They are too small to see with your naked eye. They are too small for most microscopes to see. We can only see them with really, really advanced microscopes that can cost about $1 million. Those microscopes can be used to see tiny structural details of complex molecules, like proteins. This has important implications in medical research:
These advanced microscopes can also be used to make fun videos like these:
So, an atom is small enough that we can't see it. But how small is that really? The answer is "small enough that it's really hard to conceptualize, and way too small to make it worth your while to memorize any exact numbers or figures or weird analogies that put into context how small an atom is." But, nonetheless, we will at least try to give you some concept of how small an atom is.
First, we should clarify that not all atoms are the same size. The smallest atom, hydrogen, is about 0.0000000001 meters in diameter, or 0.00000004 inches. The largest naturally occurring atom, uranium, is about 0.000000175 meters in diameter, or 0.000007 inches. Still have no idea how big this is? Lets try zooming in on the human head until we get down to an atom.
Still not helpful? Maybe this animation can explain it better.
Still not helpful? Maybe this animation can explain it better.
So... the point is, atoms are really, really small.
What are the parts of an atom?
We mentioned in the last lesson that atoms are made of of smaller particles. The main structural units of an atom are called subatomic particles, and their names are:
These different parts of an atom have unique properties, and changing how many of each type an atom has changes how that atom behaves. So, it's pretty important to understand what protons, neutrons, and electrons are and how they influence atomic behavior.
- Protons
- Neutrons
- Electrons
These different parts of an atom have unique properties, and changing how many of each type an atom has changes how that atom behaves. So, it's pretty important to understand what protons, neutrons, and electrons are and how they influence atomic behavior.
|
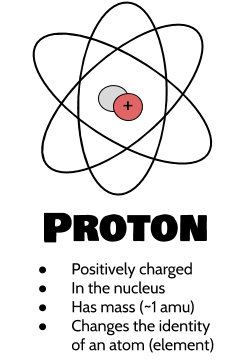
Protons are positively charged, meaning that it's kind of like one end of a magnet that will attract negatively charged things. They have a mass of about 1 amu (atomic mass unit, or a unit of mass designed to describe the weights of atoms, since they're so small. This unit is roughly defined as the mass of a proton or neutron). As we'll discuss in more detail in a moment, protons are found at the very center of the atom in a ball called the nucleus, which essentially acts as an anchor for the chemically reactive electrons, which are negatively charged (and therefore attracted to the positive electrons! Opposites attract!). Changing the number of protons changes the atomic properties so much that even adding one more or taking away one proton completely changes the type of atom something is! This is partly because it changes the number of electrons that the atom can hold onto. Elements (types of atoms) are defined by the number of protons that they have.
|
|
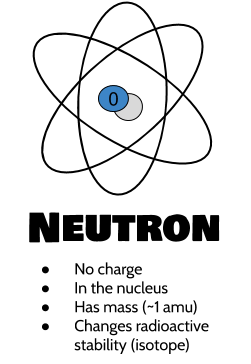
Neutrons are neutrally charged, meaning they have no net charge (they are neither positive nor negative). They are similar in size to protons and also have a mass of about 1 amu. They are also found in the nucleus of an atom. They are important for keeping the nucleus of an atom held together: Because protons are positively charged and like charges repel like charges (like matching ends of a magnet), neutrons help them protons to stay together by providing just a little bit of space between them. Changing the number of neutrons changes the isotope of that atom, which affects its radioactive stability. The element stays the same.
|
|
Electrons are negatively charged, and they orbit around the positively charged nucleus due to the electromagnetic force attracting them together. They have mass, but this mass is so small compared to the mass of protons and neutrons that we don't even count it when we're calculating the mass of an overall atom. The mass is said to be negligible, or so small that it's not important. But, that doesn't mean the electrons themselves aren't important! In fact, electrons are arguably the most important part of an atom, because they influence how that atom interacts with other atoms, which allows them to form large, complex molecules, which is how atoms become useful. We'll talk more about this when we talk about molecules and chemical bonding. Electrons also determine how atoms and molecules can behave in chemical reactions, which we'll talk about a little in this course, but that's mostly reserved for more advanced chemistry classes.
|
How are the parts of the atom organized?
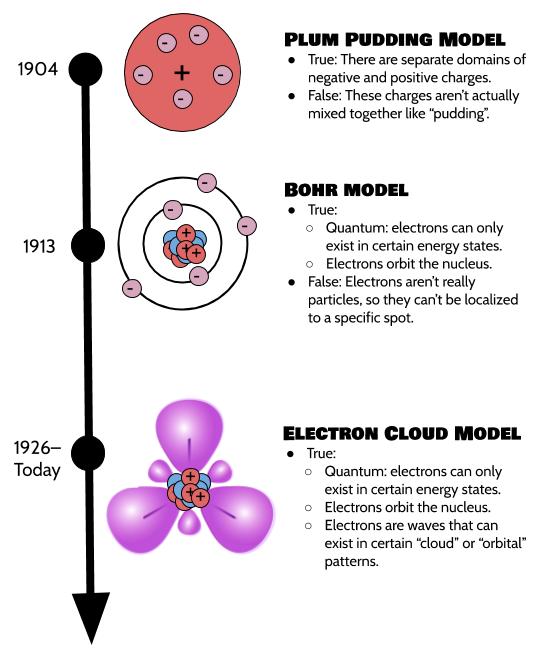
Our understanding of the structure of the atom has changed a lot with new advances in science, and it very well might keep changing as we learn new things about physics. In 1904, J.J. Thomson proposed the plum-pudding model, which said that all the parts of atoms are mixed in together.
This was debunked by the finding that electrons are actually separate from the nucleus (protons and neutrons) and can only be found in quantum "rungs," meaning that they can only have a certain fixed number of energy levels, and can't be anywhere in between. "Quantum" may seem like a really complicated idea, but it just means things can only exist in integer states, like 1, 2, 3, and not as fractions or decimals. Your family is quantum! These new scientific findings were put together and described in Niels Bohr's Bohr model, proposed in 1913.
Then, we discovered that—even though all that quantum stuff is still very true—electrons weren't even really particles at all, they're more like waves (like sound waves or light waves). So, it's not really accurate to say they're in one specific location, like a moon orbiting a planet (which was the Bohr model's idea). So, in 1926, Erwin Schrödinger—also known as “that cat guy”—proposed an electron cloud model that reconceptualizes electrons as waves that can be statistically expected to fall within a particular wave pattern. These patterns are called atomic orbitals or "clouds". This electron cloud model is the one we still use today.
This was debunked by the finding that electrons are actually separate from the nucleus (protons and neutrons) and can only be found in quantum "rungs," meaning that they can only have a certain fixed number of energy levels, and can't be anywhere in between. "Quantum" may seem like a really complicated idea, but it just means things can only exist in integer states, like 1, 2, 3, and not as fractions or decimals. Your family is quantum! These new scientific findings were put together and described in Niels Bohr's Bohr model, proposed in 1913.
Then, we discovered that—even though all that quantum stuff is still very true—electrons weren't even really particles at all, they're more like waves (like sound waves or light waves). So, it's not really accurate to say they're in one specific location, like a moon orbiting a planet (which was the Bohr model's idea). So, in 1926, Erwin Schrödinger—also known as “that cat guy”—proposed an electron cloud model that reconceptualizes electrons as waves that can be statistically expected to fall within a particular wave pattern. These patterns are called atomic orbitals or "clouds". This electron cloud model is the one we still use today.
Even though the electron cloud model is the most accurate (per our current scientific understanding), it's also more complicated than you really need to understand for this class. We will mostly use the Bohr model going forward, since this gives us enough information to be really useful but not so much that it's too complicated to understand. To make sure you have a really solid understanding of the Bohr model, we'll also go into more detail on the Bohr model in the next part of this lesson.
Summary
This video provides a good overview of the most important topics discussed in this lesson:
You should understand:
- That atoms are incomprehensibly small.
- That atoms are made up of protons, neutrons, and electrons, which have these traits:
- Protons: Positively charged, located in the nucleus, have a mass of about 1 amu, and change the atom so much that it becomes a new element.
- Neutrons: Not charged, located in the nucleus, have a mass of about 1 amu, and change the radioactive stability of an atom (its isotope)
- Electrons: Negatively charged, orbit the nucleus, have negligible mass, and changes the chemical reactivity of the atom
- That our best model of the atom is more advanced than the one we use most in this class (the Bohr model), and that models can get replaced over time as scientific understanding becomes more advanced.
Quick Quiz
Answer the following to help solidify your knowledge of what you've learned:
Content contributors: Emma Moulton and Emily Zhang