Water
There are a lot of really cool, important molecules in our bodies, but one in particular stands out as being especially important. In fact, it’s so important that it takes up 60-70% of your entire body, by weight. That molecule is water. Yep, good, old-fashioned H₂O.
Water Does a Lot of Hydrogen Bonding
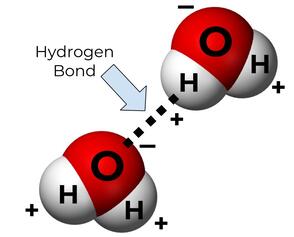
Why is water so special? It all comes back to hydrogen bonding. We’ve talked about hydrogen bonding before, but I’ll give you a quick refresher. Oxygen is an electron-hog (it’s electronegative), so it pulls negatively charged electrons away from hydrogen and toward itself. That makes the oxygen end of water relatively negatively charged, while the hydrogen end is relatively positively charged. Since opposites attract, that makes water really “sticky” (on a molecular level). It sticks to itself really well, and it dissolves most other stuff pretty well. The bond between the positive end of water and the negative end is called a hydrogen bond.
Hydrogen Bonding Gives Water Superpowers
This hydrogen bonding gives water some very unique features that make it really good at sustaining life. One would dare even say that it’s hydrogen bonding that gives water the superpower of sustaining life (which, if you ask me, is a pretty good super power.) Let’s look at the more specific superpowers that water has because of hydrogen bonding.
1. It dissolves most stuff pretty well. We learned in chemistry that water is polar (relatively positively charged on one end and relatively negatively charged on the other end). This property gives it the ability to dissolve a lot of other stuff. This is important because, in order for chemical reactions to occur, molecules need to bump into each other.
1. It dissolves most stuff pretty well. We learned in chemistry that water is polar (relatively positively charged on one end and relatively negatively charged on the other end). This property gives it the ability to dissolve a lot of other stuff. This is important because, in order for chemical reactions to occur, molecules need to bump into each other.
Chemical reactions can only happen if molecules bump into each other. Being in a solvent like water lets molecules move around so that they can bump into each other and react.
Basically, water makes it possible for molecules to swim to each other so that chemical reactions can happen. In superhero terms, water carries stuff where it needs to go:
Basically, water makes it possible for molecules to swim to each other so that chemical reactions can happen. In superhero terms, water carries stuff where it needs to go:
2. It is relatively hard to heat up and cool down. Water has a high specific heat, which means that it heats up very slowly in a hot environment and cools down very slowly in a cold environment.
Water is heat-resistant and cold-resistant
Can you imagine if your blood boiled every time it was hot out and froze every time it was cold out? That would make life impossible! Water needs to stay liquid to be a good solvent. It’s also helpful if it stays at one temperature (for us humans, that’s 98.6 °F, or 37 °C), because a lot of the chemical reactions in our bodies like to stay at one temperature. The fact that water is hard to heat up or cool down makes it a lot easier to hit that target temperature.
Lake Ontario in the summer versus the winter. Note: Still liquid.
3. It’s buoyant. Like we learned in chemistry, water has a high surface tension and is buoyant enough to prevent a lot of things from sinking. It basically has the super-strength to support a bunch of heavy stuff floating on top of it:
Water’s buoyancy (superstrength) also supports the weight of, say, the cells or our bodies to prevent them from collapsing in on each other. This helps our body to stay in a shape (doesn’t have to be “superhero shape” just “not melted shape”), instead of becoming piles of goop.
Water’s buoyant force prevents our cells from crushing in and turning us into piles of goop.
Every single one of these superpowers is linked back to water’s ability to participate in hydrogen bonding. Water dissolves stuff because it can make hydrogen bonds or ion-dipole bonds with the things it’s dissolving. It has a high specific heat because those hydrogen bonds are hard to break. And it’s buoyant because those hydrogen bonds hold water tightly together, giving it a relatively high density and surface tension. Yay hydrogen bonding!
Even More Superpowers of Water
There are also many more features of water that make it really special for all different kinds of life on earth, not just us humans. It also helps keep our environment stable. This video goes over a few of those other cool features of water, all of which come back to hydrogen bonding:
Summary
What’s the takeaway here? Water’s unique structure gives it unique properties and functions. We’ve gone into a lot of detail on this particular molecule and how it’s structure gives it those functions, because it’s an especially important molecule that you already understand pretty well from our chemistry unit. But, it’s not the only molecule that’s special because of its structure. In fact, literally every unique property of every molecule comes down to its unique structure. The different chemical reactions that can occur with a molecule come down to its structure. The ways that molecules can interact with other molecules come down to their structures. The boiling point, melting point, ability to dissolve in water, the color, everything comes down to structure. Understanding this general principle--unique structure provides unique function--is one of the key ideas in biochemistry, biology, chemistry, and really all of science.
Is anyone else really thirsty now?
You should understand:
You should understand:
- That water’s unique structure gives it unique functions, because the polar structure allows for hydrogen bonding.
- That water’s ability to hydrogen bond really well gives it unique properties that allow it to sustain life, including:
- It’s a great solvent (dissolves stuff well).
- It’s has a high specific heat (is hard to heat up and cool down)
- It has a high buoyancy (it is able to support a lot of weight and keep cells from collapsing)
- It’s a great solvent (dissolves stuff well).
- That the unique structure of everything gives it unique functions.
Learning Activity
Content contributors: Emma Moulton